Why Detox?
Detox Navigation
Detox Program: A Total Body Cleanse
Detox 1: Colon Detox & Intestinal Cleanse
Detox 2: Heavy Metal Cleanse
Detox 3: Kidney Detox
Detox 4: Liver Detox
Detox 5: Blood Detox
Detox Library
Clinically Proven Oral Chelation
By: Jon Barron
Heavy Metals are becoming such a major problem today that finding a formula that removes them has always been one of my top priorities. In 2003, I created an organic, oral chelation formula for Baseline Nutritionals®. It’s one of my favorite formulas, but other than a handful of doctors who each regularly use nearly 100 bottles a month in their practices, very few people were aware of the power of this formula the first two years it was available. But, with the release of the clinical studies in 2005 proving its effectiveness, that changed.
Clinical studies completed in September 2005 prove that it’s possible to naturally remove an average of 87% of Lead, 91% of Mercury, and 74% of Aluminum from the body within 42 days. The study was conducted by healthcare professionals at The Optimal Wellness Test Research Center in Nevada (not affiliated with Dr. Mercola’s Optimal Wellness Center in Ilinois).
The problem with heavy metals is that they accumulate in the body causing numerous health problems that can seriously impact literally every major organ in the body. Studies show that heavy metals in the body may be implicated in everything from Alzheimer’s to cardiovascular disease, from behavioral problems to kidney dysfunction, Parkinson’s disease, epilepsy, and even paralysis. The results of the clinical study on the formula should certainly be of interest to those looking for a proven, natural, affordable method for removing damaging heavy metals from the body.
Oral Chelation Study Summary
The study involved fifty subjects with various beginning wellness levels, age, exercise levels, race, sex, and health levels. Subjects were first tested with clean bodies — meaning they consumed no prescription drugs, supplements, nor special diets for 1 week prior to their first use of said formula. Strict guidelines were followed during the study such as restrictions in using any other supplements, OTC drugs, or from altering their diets.
The study also showed that nitrogen, nitrates, and ammonias increased overall to wellness numbers by 25%. This means that the product also decreased other toxic levels that are stored intra-cellular in the body. These toxins are often the cause of very low pH numbers that are difficult to move into normal wellness range. But while on the cilantro and chlorella formula, pH ranges moved an average of 33% into the wellness range. Eliminating the stored toxins and heavy metal toxins allows the cellular body to remove stored acids and other unbalancing fluids.
The researchers concluded that an organic herbal oral chelation formula of cilantro and chlorella is a very effective heavy metal chelator and a great product for those that have very acidic pH intra-cellular imbalances.
The Herbal Oral Chelation Formula
The formula is a tincture of cilantro and chlorella.
Why cilantro and chlorella?
 Because cilantro changes the electric charge on intracellular deposits of heavy metals to a neutral state, which relaxes their tight bond to body tissue, freeing them up to be flushed from the body. Studies have shown that levels of mercury, lead, and aluminum in the urine increase significantly after consuming large amounts of cilantro.1 It seems that cilantro changes the electric charge on intracellular deposits of heavy metals to a neutral state, which relaxes their tight bond to body tissue, freeing them up to be flushed from the body2 — exactly the results seen in the Clinical Study.
Because cilantro changes the electric charge on intracellular deposits of heavy metals to a neutral state, which relaxes their tight bond to body tissue, freeing them up to be flushed from the body. Studies have shown that levels of mercury, lead, and aluminum in the urine increase significantly after consuming large amounts of cilantro.1 It seems that cilantro changes the electric charge on intracellular deposits of heavy metals to a neutral state, which relaxes their tight bond to body tissue, freeing them up to be flushed from the body2 — exactly the results seen in the Clinical Study.
Once free, the next step is to actually facilitate the removal of the metals from the body. And here’s where chlorella comes in. Chlorella possesses the capacity to absorb heavy metals. This property has been exploited as a means for treating industrial effluent that contains heavy metals before it is discharged, and to recover the bio-available fraction of the metal in the process. In studies undertaken in Germany, high doses of chlorella have been found to be very effective in eliminating heavy metals from the body – from the brain, intestinal wall, muscles, ligaments, connective tissue, and bone.3
Together, these herbs create a powerful oral chelation formula.
The bottom line is that this formula is now proven to be a major weapon in the fight against heavy metal toxicity. Once cleaned out after 42-days, a two week cleanse every 3-4 months should be enough to keep you relatively metal free – unless you eat a lot of high-mercury fish, or have more than a few amalgam fillings, in which case every 2 months is advisable. Incidentally, you do not want to use a chelation formula every day. Your body actually needs small amounts of some heavy metals.
Heavy Metal Toxicity
Lead
With the elimination of lead-based house-paint, and the increased use of lead-free gasoline, lead poisoning is certainly less prevalent. However, low-level toxicity is still an issue. Coal burning power plants still spew lead into the atmosphere, and like mercury and aluminum, the problem with lead is that it accumulates, unless you take active steps to remove it. The EPA estimates that 10 to 20 percent of human exposure to lead may come from lead in drinking water. Infants who consume mostly mixed formula can receive 40 to 60 percent of their exposure to lead from drinking water. The EPA warns that if lead is not detected early, children with high levels of lead in their bodies can suffer from damage to the brain and nervous system, behavior and learning problems (such as hyperactivity), slowed growth, headaches, and more. However, adults are still at risk and can suffer from reproductive problems (in both men and women), high blood pressure, digestive problems, nerve disorders, memory and concentration problems, and muscle and joint pain.
Aluminum
As for aluminum, it has been known for 20 years that once it enters your body, it accumulates in your brain, where it kills off neurons, leading to memory loss. And thanks to the significant amounts of aluminum found in food emulsifiers, antiperspirant deodorants, hair sprays, baking powder, many types of toothpaste, much of our drinking water, and most of our cookware, you are exposed to a lot of aluminum over the course of your life. There has been much speculation, therefore, that aluminum may be one of the prime factors in the onset of Alzheimer’s disease. The connection between aluminum and Alzheimer’s disease became even stronger when in 1995, Neurotoxicology reported that the widespread use of aluminum salts to purify water could account for the large numbers of people suffering from Alzheimer’s.
And recently, the final piece of the puzzle may have fallen into place: the connection between aluminum and fluoride. New research has revealed that fluoride in drinking water makes the aluminum that we ingest more bio-available. As was reported in Brain Research, Vol.7 84:98, the combination of aluminum and fluoride causes the same pathological changes in brain tissue found in Alzheimer’s patients.
Note: there is a significant difference between metallic aluminum and plant-derived aluminum, which is in the form of aluminum hydroxide. No studies have ever shown a connection between aluminum hydroxide and toxic levels of aluminum in the human body — which is a good thing, because there is a lot of it in our food supply.
Like mercury, the danger from lead and aluminum is not the result of large doses, but the result of a steady accumulation over years — as they do not easily clear from the body unless you take conscious steps to remove them.
Mercury, Deadly Beauty
 People have known about the dangers of mercury since the days of the Roman Empire, when slaves who worked in the “quicksilver” mines died horribly after 2-3 years exposure. And in the 19th century, the workers who used mercury to make hats went bald and suffered from severe muscular tremors, dementia, and fits of wild, uncontrollable laughter. Thus the phrase: “Mad Hatter.”
People have known about the dangers of mercury since the days of the Roman Empire, when slaves who worked in the “quicksilver” mines died horribly after 2-3 years exposure. And in the 19th century, the workers who used mercury to make hats went bald and suffered from severe muscular tremors, dementia, and fits of wild, uncontrollable laughter. Thus the phrase: “Mad Hatter.”
It’s no secret that mercury is one of the most toxic metals known. Numerous studies have shown its impact on health. There is strong evidence that mercury lowers T-Cell counts. This, alone, implicates it in cancer, autoimmune diseases, allergies, Candida overgrowth, and multiple sclerosis. In fact, due to other studies that showed mercuric chloride increased several types of tumors in rats and mice, and methyl mercury caused kidney tumors in male mice, the EPA has determined that mercuric chloride and methyl mercury are possible human carcinogens. It has also been shown that mercury cuts the oxygen carrying capacity of blood by half. This would account for many instances of chronic fatigue.4
Mercury also has an affinity for brain tissue and is implicated in brain tumors and dementia. And, finally, mercury has an affinity for fetal tissue, which accounts for its implication in birth defects. In 2002, the National Academy of Sciences found strong evidence for the toxicity of methyl mercury to children’s developing brains, even at low levels of exposure. A recent study from the Centers for Disease Controls found that as many as 637,233 American children are born each year with mercury levels of more than 5.8 µg/L (5.8 micrograms per liter), the level associated with brain damage and loss of IQ.
Today, we face two primary sources of exposure: our food supply and our dental fillings.
48 Tons of Toxins in Our Food and Water
There is nothing complex about the process. Mercury is a naturally occurring toxin, which is found in soil, rocks, wood, and fuels like coal and oil. Simple soil erosion deposits mercury in rivers and lakes, but concentrations remain low, unless, as has been discovered in the recently deforested regions of the Amazon, erosion reaches extraordinary levels. The burning of rainforests also releases mercury that has been taken up from the soil by the trees.
But the major source of mercury in our food chain, responsible for about 1/3 of the levels found in our bodies, is our burning of coal to generate electric power. That is the single greatest contributor to the problem. Mercury that naturally occurs in the coal is released during burning and enters the air; it is then precipitated into the oceans, lakes, and rivers by rain. According to the EPA, coal-fired power plants in the United States emit about 48 tons of mercury into the air every year — and more than half of this mercury falls within 5 miles of the plant itself. When it reaches the water, microorganisms consume it and convert it into a substance called methyl mercury.
Mercury In the Food Chain
A study at the University of Tennessee recently rated methyl mercury among the most dangerous poisons on Earth (just behind plutonium). It has no known beneficial use in the body, and it accumulates in the muscle tissue of fish, animals, and humans. When minnows eat plankton or algae that is contaminated with methyl mercury, it is deposited in their flesh; larger fish prey upon the minnows, and the toxin travels straight up the food chain to the most prized game fish — the big predators like bass, pike, walleyes, brown trout; and to all the finest food and sport fish of the seas — tuna, swordfish, shark, roughy, marlin, and halibut. According to the EPA, fish at the top of the aquatic food chain bio-accumulate methyl mercury to a level approximately 1 million to 10 million times greater than dissolved concentrations found in surrounding waters.
Of course, when you climb one more rung up that food chain, you find us, the people who eat fish. Just like the predatory fish that we catch and eat, we store mercury in our tissues. Just like the ancient Romans, we know that high exposure to mercury is fatal. But…
Mercury Rising
In 1997, the EPA under the Clinton administration presented a detailed study that revealed the hazards of mercury contamination, pinpointed coal-fired power plants as the leading source of emissions, and promised action. But nothing was done. The EPA had begun work on a plan to address mercury pollution in December 2000 and in a 2001 presentation, the agency said that 90 percent of mercury emissions from coal-fired power plants could be cut, using what is known as the Maximum Achievable Control Technology (MACT), by 2008.
More recently, the Bush administration decided that the coal-fired power industry would be exempt. Therefore, levels will continue to climb — not fall.
Mercury Fillings
The American Dental Association has resolutely maintained for years “when mercury is combined with the metals used in dental amalgam, its toxic properties are made harmless.” If this were true, it would be miraculously fortuitous.
Dentists have used amalgam, which consists of mercury, silver, tin, copper, and zinc, for several hundred years. Here in the United States, it made its appearance in the early 1800s.
From the beginning, there were a number of dentists who were concerned by the presence of mercury, since by that time it was fairly well known that mercury was poisonous. In fact, these concerns were so strong, that by the mid-1940s, several dental societies, including the American Society of Dental Surgeons, had joined together to stop the use of amalgam fillings. But the problem all along has been that amalgam is just too easy to work with, and whatever ill effects people experience are too far down the road to matter; so dentists, as a group, have fought for its continued use. And, in fact, the American Dental Association was founded in 1859 — primarily to promote the use of mercury amalgam as a safe and desirable tooth filling material. There were no tests done. No studies. Nothing! Amalgam was promoted because it was easy to work with. The reason mercury was used in it was because mercury serves to “dissolve” the other metals and make a homogenous whole.
The early position of the ADA was that mercury reacts with the other metals to form “a biologically inactive substance” so that none of it ever makes its way into your body.
Unfortunately, numerous studies conducted in the 1970s and 80s proved conclusively that the mercury from fillings (primarily from mercury vapor created when you chew) makes its way into your body, ending up in your lungs, heart, stomach, kidneys, endocrine glands, gastrointestinal tract, jaw tissue, and brain.
Once it became irrefutable that mercury from the fillings was ending up in your body, it then became mandatory that the ADA find a new defense. Again, not based on study, it became the position of the ADA that: Well yes, maybe some mercury does make its way into your body, but at levels that are so low it has no effect on your health. Unfortunately, that’s just not true either. Like so many other toxic substances, the real problem with mercury is that it is a cumulative poison. The body holds onto a significant percentage of the mercury that enters it.
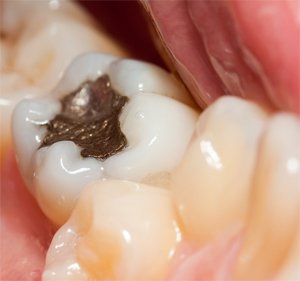
Note: There is no safe way to remove amalgam fillings. Recent studies showed that even with strong air and water suctioning, water rinses, and a rubber dental dam, significant amounts of mercury were later found in the individual’s lungs, kidneys, endocrine organs, liver and heart, whereas no mercury was detected in those tissues prior to removal of the fillings.
Since the government is not going to help limit your exposure to mercury in your food supply, and since your dentist and the ADA are not going to limit your exposure in your amalgam fillings, there is only one place you can turn for help — yourself. You need to regularly cleanse accumulated mercury from your body.
The Bottom Line on Heavy Metals
When it comes to heavy metals, there are three clear steps available to you.
- Avoid exposure. Say no to new amalgam fillings and, if possible, have a dentist who understands the process replace your existing fillings. (However, you will need to detox after removal.) Avoid aluminum cookware and aluminum based deodorants. Stop eating high-mercury fish such as swordfish, shark, roughly, and albacore tuna. And filter fluoride and lead out of your drinking water.
- Regularly sweep heavy metals from your colon and draw them from the tissue lining the walls of your intestinal tract using a powerful herbal colon detoxifier. (Note: colonic irrigation will not remove heavy metals from your intestinal walls.)
- Routinely cleanse heavy metals from your body — since they will not leave on their own. And now, thanks to this chelation formula, you have an easy, no fuss, inexpensive way to do just that.
Addendum – 6 April 2013
For some years now, the heavy metal formula described above has been extremely popular. And why not? It’s clinically tested and people love the results. Anecdotally, we’ve seen a steady stream of testimonials. Nevertheless, through the years, there have been two recurring problems experienced by a small percentage of people.
- Some people complained of getting slightly “spacey” while using the formula.
- And some people who relied on urine provocation testing to determine how well the formula worked for them would freak out when they saw higher numbers after using the formula—even though that would be expected and was a sign that metals were actually pulled out of the soft tissue and clearing the body.
As it turns out, these problems are related. What’s happening is that the cilantro is unbinding and freeing up the heavy metals—particularly mercury—faster than the chlorella can escort it from the body. Eventually, the chlorella catches up, but until then, the temporarily higher levels of metals in the blood can make you feel a bit woozy. Also, this lag time will cause the numbers seen in provocation testing to remain higher for longer periods of time.
So, the question arose, was there any way to enhance the formula that could help at least mitigate these problems, understanding that they can never be totally eliminated since they are inherent in the detox process?
Humic and Fulvic Acid
Humic acid, fulvic acid, and humin are types of humic substances or humates that are the major components of organic matter found throughout nature as a result of the action of millions of beneficial microbes on decomposing plants—a process known as humification. Humic acid and fulvic acid are considered to be the chemically bioactive compounds. They are black or dark brown in color and are of high molecular weight. Humins are the brownish amorphous substance that is produced from acidic reactions on certain sugars, carbohydrates, or the remaining residue of humate extraction and are considered less biologically active.
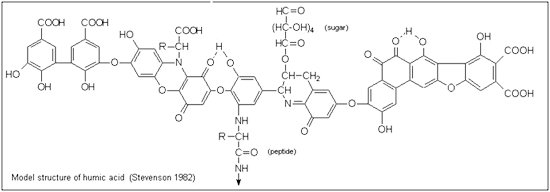
Because humic and fulvic acid are actually large, heterogeneous collections of polyaromatic macromolecules, and because they are so complex and their sourcing components (the decaying plant and animal matter) so variable, their molecular structures are not precisely known—but are, rather, generalized.5 The hypothetical structure for humic acid, for example, contains free and bound phenolic OH groups, quinone structures, nitrogen and oxygen as bridge units, and COOH groups variously placed on aromatic rings.
The hypothetical model structure of fulvic acid contains both aromatic and aliphatic structures, both extensively substituted with oxygen-containing functional groups.
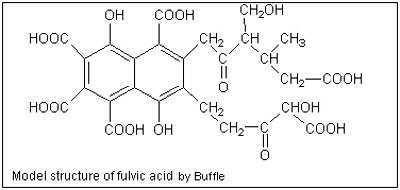
In addition, beyond their core structures, both humic and fulvic acid incorporate other molecules into their structure such as protocatechuic acid, vanillic acid, vanillin, resorcinol, ferulic acid, benzoic acid, and other similarly structured polyphenols resulting from the breakdown of the structural lignans in plant cell walls. It’s also important to note that the exact composition of humic and fulvic acid will vary according to its source. Different sources produce humic/fulvic acid of varying efficacy.
As you may have guessed from the description so far, any time you buy a humic or fulvic acid formula for human use, you’re actually getting a mix of all three components. Theoretically, if something is called fulvic acid VS humic acid, it is merely indicative of the ratios involved in that blend. In other words, something called fulvic acid would have a higher proportion of fulvic acid than something labeled humic acid. Humic acid is naturally dominant, but the fulvic acid component can be concentrated during extraction. This is important since fulvic acid has more chelating potential than humic acid.
It is actually possible to obtain “organic” humic and fulvic acids. To call something “organic” in the U.S. requires approval by the National Organic Standard Board NOSB. In fact, it is possible to track down suppliers of high quality fulvic acid that not only matches the requirements of the USDA, but also of the European Union, and the Japanese Organic Standard Board. In addition, although there are humic and fulvic acids everywhere on the surface of the earth, only large deposits created via biotic and abiotic factors over 60 million years ago are of commercial value. In the U.S., such deposits are buried several feet below ground in pristine ancient lake beds that are known to be untouched by man until recently. These deposits are known to contain absolutely no heavy metals and were not exposed to GMOs. Thus, they’re qualified to be classified as organic or natural and suitable for organic food production. Such deposits are also a rare find in the fulvic and humic acid industry.
Fulvic Acid and Chelation
 There are many health benefits associated with the use of supplemental humic and fulvic acids, including:
There are many health benefits associated with the use of supplemental humic and fulvic acids, including:
- Control of inflammation
- Stimulating metabolism
- Working as powerful free radical scavengers
- Regulating the thyroid and thymus glands
- Oxygenating the blood
- Maintaining optimum alkalinity
But the one we are concerned with today is their ability–and especially that of fulvic acid—to chelate toxic and/or inorganic metals and escort them safely out of the body.
The word “chelate” itself is derived from the Greek word chela, which refers to the pincer-like claw of a crustacean or arachnid, such as a lobster, crab, or scorpion. The term chelate, then, suggests the way in which an organic compound grabs onto the cation elements (in this case, toxic heavy metals) and carries them to the liver for processing and out of the body through the urine or feces.
In order for a compound to be called a true chelating agent, it must have certain chemical characteristics. It must have at least two sites capable of donating electrons to the metal it chelates. For true chelation to occur, the donating atom(s) must also be in a position within the chelating molecule so that a formation of a ring with the metal ion can occur. Humic and fulvic acid both qualify as true chelating agents.6
Humic/fulvic acid has been extensively tested for its ability to chelate toxic heavy metals from the body. For example, in one study, oral consumption of humic/fulvic acid administered daily for six weeks significantly decreased blood cadmium levels and increased urine cadmium in 31 adult workers continuously exposed to occupational cadmium.7 In the majority of subjects, initial abnormally low serum iron levels increased, and markers of kidney and liver function improved. In other studies humic/fulvic acid has been shown to decrease the absorption of heavy metals in the intestinal tract and reduce their toxic effects.8
Other studies have indicated that humic/fulvic acid may also be beneficial in helping your body to rid itself of lead, strontium, and mercury.
Adding Fulvic Acid to the Existing Formula
The problem in enhancing any formula is that you can’t just simply add something to the formula. There’s only so much room you can work with. In a 2 oz bottle, there’s only 2 ounces. If you add something, by definition, you have to take something out to make room for it, and if that something is a powerful bioactive, you have to be concerned that you’re not losing more than you’re gaining. On the other hand, in rare cases, you can find an ingredient that can be added that doesn’t require you to remove an “active” ingredient. As it turns out, we could make room for adding a sizeable amount of pristine, organic, liquid humic/fulvic acid by dropping out some of the alcohol and water base in the tincture since they play no active role in the heavy metal chelation formula. Thus, we get to add a large amount of a powerful chelating agent to the formula without reducing the amount of bioactive cilantro or chlorella even one microgram. In formulation, this has to be considered the ultimate win/win scenario.
Humic/Fulvic Conclusion
When we tested the revised version of the heavy metal chelation formula with the added fulvic acid solution, the first thing that we noticed was that several people who had previously complained about feeling “off” or “spacey” when using the formula no longer had that complaint. They felt no dizziness or wooziness—indicating that the fulvic acid was working as expected. And under limited urine provocation testing, the numbers for heavy metals in the blood began to drop much more quickly in the days after using the formula than with previous versions. By all indications, the efficacy of the formula has been significantly enhanced.
In terms of color, smell, and taste, the new version is certainly interesting. There’s no denying that it’s now dark brown, bordering on black. Get any on your fingers and it temporarily stains them. You have to wash it off with soap and water; it won’t just rinse off. As for smell, the aromatics in it, such as the vanillin, now give the formula a slightly sweet, earthy smell—almost like a rich loam soil laced with vanilla and spices. It’s not strong, and it’s not offensive. Consider it nostalgic—taking you back to your days on the farm…or at least in your patch of your local victory garden. As for taste, it’s actually quite mild, surprisingly–when mixed with even a small amount of juice–almost non-existent.
In other words, because of the color, it’s recommend that you mix it with 1-2 oz of a dark juice such as grape or pomegranate as opposed to something light and colorful like orange juice. The taste when combined with orange juice is actually quite okay, but somehow drinking black orange juice is disconcerting.
It’s also recommended that you drink a glass of water after consuming a 4 mL serving of the formula.

- 1.Omura Y, Beckman SL “Role of mercury (Hg) in resistant infections & effective treatment of Chlamydia trachomatis and Herpes family viral infections (and potential treatment for cancer) by removing localized Hg deposits with Chinese parsley and delivering effective antibiotics using various drug uptake enhancement methods.” Acupunct Electrother Res. 1995;20(3-4): 195-229. http://www.ncbi.nlm.nih.gov/pubmed/8686573
- 2.Omura Y, Shimotsuura Y, Fukuoka A, Fukuoka H, Nomoto T. “Significant mercury deposits in internal organs following the removal of dental amalgam, & development of pre-cancer on the gingiva and the sides of the tongue and their represented organs as a result of inadvertent exposure to strong curing light (used to solidify synthetic dental filling material) & effective treatment: a clinical case report, along with organ representation areas for each tooth.” Acupunct Electrother Res. 1996 ;21(2): 133-160. http://www.ncbi.nlm.nih.gov/pubmed/8914687
- 3.Klinghardt, D. “Amalgam/Mercury Detox as a Treatment for Chronic Viral, Bacterial, and Fungal Illnesses.” Explore. Volume 1997;8, No 3.
- 4.Mattingly RR, Felczak A, Chen CC, McCabe MJ Jr, Rosenspire AJ. “Low concentrations of inorganic mercury inhibit Ras activation during T cell receptor-mediated signal transduction.” Department of Pharmacology, Wayne State University, Detroit, Michigan 48201, USA. Toxicol Appl Pharmacol. 2001 Nov 1;176(3):162-8. http://www.ncbi.nlm.nih.gov/pubmed/11714248
- 5.A. Piccolo (2002). “The Supramolecular structure of humic substances. A novel understanding of humus chemistry and implications in soil science”. Advances in Agronomy 75: 57–134 http://www.sciencedirect.com/science/article/pii/S0065211302750037
- 6.D. Gondar, R. López, S. Fiol, J.M. Antelo, F. Arce. “Cadmium, lead, and copper binding to humic acid and fulvic acid extracted from an ombrotrophic peat bog.” Geoderma.2005.12.003. http://www.researchgate.net/publication/223954546_Cadmium_lead_and_copper_binding_to_humic_acid_and_fulvic_acid_extracted_from_an_ombrotrophic_peat_bog
- 7.Hudák, A., et al. “Effect of the consumption of humic acid with bound complex micro elements in cases of occupational cadmium exposure.” Central European Journal of Occupational and Environmental Medicine, 1997. 3(3): p. 175-186. http://www.omfi.hu/cejoem/Volume3/Vol3No3/ce973-2.HTM
- 8.Glynn AW. “Fulvic and humic acids decrease the absorption of cadmium in the rat intestine.” Arch Toxicol. 1995;70(1):28-33. http://www.ncbi.nlm.nih.gov/pubmed/8750902
Interested in a Full Body Detox?
107 Comments
Submit a Comment Cancel reply
You must be logged in to post a comment.

Will your chelation formula
Will your chelation formula remove iron? I have hemochromatosis and very high iron levels. Phlebotomy is becoming more and more problematic. Can you help me? Thank you, Maureen
No, both chlorella and
No, both chlorella and cilantro actually contain iron, so they are not ideal for removing it from the body. Check with your doctor, but in general, the best solution for lowering iron levels is to donate blood.
Inositol hexaphosphate (IP6)
Inositol hexaphosphate (IP6) removes iron (and other minerals) from the body. It should be taken on an empty stomach. Don’t know if it will help with hemochromatosis – you’ll have to investigate that issue yourself.
The video of making tinctures
The video of making tinctures yourself is quite dangerous. I am a naturopath and you must understand the active constituents that you are wanting to extract from your plant. Also understanding the full genus and species and the chemotype. Alcohol only extracts certain constituents and you you need properly managed conditions to ensure adequate manufacturing process.
How can I make it myself – a
How can I make it myself – a tincture of cilantro ?
And what else, if I want to make it myself, amounts of tincture and chlorella…?
I am extremely sensitive and
I am extremely sensitive and have difficulty detoxing (genetic and lots of history). I know I have candida and heavy metals, especially mercury. I am chronically constipated so my times on chlorella have not been workable.
How would you deal with this situation?
Thanks.
Lynne
I used IP6 to control my iron
I used IP6 to control my iron levels and it worked well for me keeping them in the normal range. I also detox which helps to remove iron.
Good day. I have
Good day. I have hemochromatosis and have recently had my mercury fillings removed. As I want to detox my body can you tell me if it advisable to use folvic acid to do this. I am concerned it might raise my iron levels Can you give me any insight you may have on this. Or if you have a contact email address for some one who might be able to answer my question would be most useful. my email address is [email protected]
I understand the logic behind
I understand the logic behind the treatment and so started to take chlorella, however, as a result I suffered from vomits and diarrhoea. Ias this part of the process of eliminating heavy metals from the system?
If you want to learn how to
If you want to learn how to make tinctures, there’s actually a very good video on youtube called How to Make Herbal Tinctures. http://www.youtube.com/watch?v=4dloPF1QDuw. As for releasing the exact formula that Jon Barron uses to make Metal Magic, I’m sure you can understand why we would not be able to get that information from Jon.
We answered that earlier in
We answered that earlier in the chain of comments:
“No, both chlorella and cilantro actually contain iron, so they are not ideal for removing it from the body. Check with your doctor, but in general, the best solution for lowering iron levels is to donate blood.”
EDTA IV chelation certainly
EDTA IV chelation certainly is the strongest form of heavy metal detoxing, but is indeed expensive and takes a number of repeat, time-consuming visits. With the oral formula that Jon created, the initial detox recommended is six bottles, which when used at the recommended rate of four droppers, three times a day, takes about 30 days—give or take. After that, Jon recommends a maintenance program of 2 bottles every six months.
That said, the Baseline of Health Foundation does not actually have or sell any products—even Jon Barron’s formulas. We are purely an online information resource. All of Jon’s formulas, however, are available from Baseline Nutritionals.
Where can I get this and how
Where can I get this and how much does it cost?
Those results listed above
Those results listed above are virtually impossible to achieve. I would like to know how you came to the conclusion that you removed so much metal in such a short time.
As is true with virtually any
As is true with virtually any food, some people are allergic to chlorella. You may not tolerate chlorella well. Try starting with smaller amounts and see if you can build up.
It all depends on what is
It all depends on what is causing the constipation. If it’s from the sold matter in the chlorella binding you up, then Jon’s Metal Magic tincture would not be a problem, since it’s a pure liquid extract – no solid matter in it. If the problem is caused by an allergic reaction to the chlorella, however, then it wouldn’t matter what form the chlorella was in.
Will this remove cobalt?
Will this remove cobalt?
As a heavy metal, cobalt is
As a heavy metal, cobalt is likely to be removed in any chlelation process at about the same rate as lead.
All of Jon Barron's
All of Jon Barron's recommended formulas are in the chart on the page at top called "Products." If you scroll down that page, you will see a list of formulas and companies that make them. Enjoy!
Do u have like a shopping
Do u have like a shopping list of everything one would need to complete the full body cleanse that I can just go to instead of having to disect the whole web sight?
Actually, we didn’t come to
Actually, we didn’t come to that conclusion. Fenestra Research, using their Optimal Wellness Testing methodology, did. Since they ran the tests independently, you can contact them directly to find out exactly how they came to their results. http://fenestraresearch.com/ I will say the results were the best they had seen in the industry.
How can I obtain a copy of
How can I obtain a copy of the full study? Thanks! -JS
I have read many times that
I have read many times that you shouldn't do a metal detox while you still have amalgam fillings in teeth. That cilantro can take mercury into the brain. Is this true??
Trying to see reply to Toxic
Trying to see reply to Toxic girl
Please tell me what I need to remove heavy metals from my body.
Thank you,
Donna
If you go to our Detox
If you go to our Detox Program, you will see the list of products and diet plan you will need for the entire detox. Baseline Nutritionals also sells two detox kits that will get you 90% there if you plan on doing the FULL body detox that includes detoxing the colon, liver, blood, kidneys, gallbladder, etc. and heavy metals from organs. They must go in this order, which is explained in Jon's book (included in kit one).
Kit One: http://www.baselinenutritionals.com/products/intestinal-detox-package.php
Kit Two: http://www.baselinenutritionals.com/products/kidney-liver-detox-package.php
Considering that 95% of all
Considering that 95% of all people who do metal detoxing have not had their fillings removed, if what you say is true, there would be many, many stories of brain damage resulting from metal detoxing – but there aren’t.
Let’s think this through for a moment.
When you do a metal detox, the metal isn’t coming from your fillings, it’s coming from the soft tissue in your body. In other words, the detoxing is pulling out the mercury that is already in your body, not adding mercury to it. If anything, it is lowering levels of mercury in your body and brain, again, not adding to it. Make sense? Also, Jon recommends using a combination of cilantro and chlorella to even further minimize any risk. The cilantro breaks the electrical bond that the metals have with your soft tissue; and the chlorella then “sponges” up the chlorella that has been released. The bottom line is that pulling out amalgam fillings before a metal detox is not necessary. Simply repeat the detox slightly more often because you have a guaranteed source of mercury still in your mouth that is continually depositing new mercury in your soft tissue.
Hi, will this help to remove
Hi, will this help to remove copper?
Found out i am very high in copper.
The full study exists as raw
The full study exists as raw data, so it’s not particularly intelligible when seen in that form. It also contains proprietary information, which also precludes it from being published in unedited form.
As a heavy metal, copper is
As a heavy metal, copper is likely to be removed in any chlelation process at about the same rate as lead.
I am a 43 year old female
I am a 43 year old female that lives in the south. In 2007 I was diagnosed with Lupus and Sjogrens. I’ve had numerous complications since then. I am currently virtually non-functional, I can’t find my way around my city or to a doctor’s office I’ve already visited. A neurologist ordered several tests (cognitive, driving and blood). The blood test showed my manganese levels to be 9.7 (
Having a study that proves
Having a study that proves your Magic Metal chelating kit achieves extra-ordinary results well over virtually any other chelating option in the market and keep it unaccessible to all of us your potential clients makes absolutely no sense!!!
My question to you Mr Barron is…
Why you have not taken steps to convert all the raw data existing for said study into a easy to understand scientific document to support the extra-ordinary effectiveness of your Heavy Metal Detox Kit?
And if you are on the process of creating such document… would you send me a copy?
(meanwhile I’ll be patient as a client-to-be)
If you follow the proper
If you follow the proper order in detoxing, then you are not releasing toxins into the bloodstream, with the exception of heavy metal detoxing. If you start with colon detoxing, it is doing most of its work in the colon, not the bloodstream. It prepares the body for easy elimination of toxins released in the rest of the detoxes. The kidney flush doesn’t actually release toxins. It helps flush out and repair the kidneys. The sludge that is flushed out never enters the bloodstream. It is dumped into the ureters, then the bladder, and then into the toilet. It also helps soften stones in preparation for the liver cleanse. The liver cleanse purges waste from the liver into the gallbladder and then out through the colon. The toxins only enter the bloodstream if the elimination channels are plugged and there is nowhere for the toxins to go. But if you do things in the proper order, that’s not an issue. In any case, there is no harm in doing things slowly to build up your comfort level. And, as always, if you are under a doctor’s care for your liver and kidneys, then you will need to work with them in regard to doing any detoxes.
No, those metals were not
No, those metals were not specifically tested. As to a possible negative synergistic effect from multiple heavy metals, Jon explained one such example when he wrote about how fluoride makes aluminum more bio-available and better able to cross the blood brain barrier.
Remember, Jon’s heavy metal
Remember, Jon’s heavy metal detox formula is made with cilantro, chlorella, and fulvic acid. Were you instructed not to eat any of those? That would be unusual considering that chlorella and fulvic acid are recommended treatments for Candida. They also bind with heavy metals so that they can’t return to the brain from where the cilantro released them (cilantro crosses the blood/brain barrier. And keep in mind, there’s nothing to stop you from doing a program to bring Candida levels down before doing the heavy metal detox. In any case, if you start the heavy metal detox, you might want to start slowly with one dropper a day and then build up gradually over a couple of weeks to full dosage. If you have any problems, you can always back things down to a level that was previously comfortable and hold at that level. With the heavy metal detox, it’s not how much you take at one time—but how much you take over time.
Also, when it comes to Hashimoto’s disease, you might find the following article useful. http://www.jonbarron.org/article/endocrine-system-thyroid-and-parathyroid-gland
hello, can this product
hello, can this product remove coloidal silver ?, Thanks
Hi, so you do have
Hi, so you do have recommendations for the tinctures but you don't have a purchasable formula for sale, do you? Thanks!
Actually, the use of an
Actually, the use of an alcohol/water mix, which is what the video calls for, is the standard for making herbal tinctures. Of course, there are things that can be done by experts in the preparation of the water and alcohol used in the solvent mix that can make it much more effective as a solvent, but not less dangerous. So unless someone is making tinctures out of questionable herbs, there is nothing in the video that is particularly dangerous.
For obvious legal reasons, we
For obvious legal reasons, we cannot diagnose or prescribe for specific medical issues – merely provide information. With that in mind, you might find the following report useful: http://www.jonbarron.org/detox/barron-report-heavy-metal-cleanse-herbal
The current formula, which
The current formula, which contains humic/fulvic acid is not the same as the one that was originally tested. At this point, the old data is merely suggestive and no longer directly applies.
Several years ago, I tried
Several years ago, I tried Jon Barron’s formula, Metal Magic. It was not a good experience for me. I had several alarming side effects (I felt “buzzing” in my head for one). I returned the bottles for a refund. Now I’m wondering… if I cannot take the Metal Magic, what else can I do to detox heavy metals from my system? I have read that cilantro and chlorella can detox heavy metals. If I try that, how much of each should I take? Thank you for your response.
As asked, the question is not
As asked, the question is not meaningful. There are two facts you need to separate before you can answer your question.
First colloidal silver is merely the vehicle for carrying silver ions into your body. It is not a “type” of silver. Once absorbed inside your body, the silver ions separate from the water carrier and are no longer a colloid suspension.
Second silver is a metal so it is chelatable
Given that, we can rephrase your question into three parts, and arrive at a more meaningful answer.
If you take colloidal silver and a chelating agent at the same time will the chelating agent absorb the silver and prevent it from working.
And the answer is yes it is likely to absorb some of it, but the amount of silver that you are taking is likely to overwhelm the chelating agent so that most of the silver will still be absorbed by the body.
Will a chelating agent pull silver out of the body?
Yes, over time. It is likely to pull some silver out of the body.
Will it reverse argyria, the blue/grey coloration of the skin that has been known to appear in some cases where people overdo colloidal silver?
There are stories on the net about people who have used chelating agents to reverse argyria, but they are unconfirmed. Also, you need to keep in mind that if you have argyria, you are no longer talking about trace amounts of silver in your body. The silver levels are quite high and would probably require intravenous EDTA chelation if there was any chance of reversing the argyria in a relatively short time. But without any testing or studies to support it, that is merely speculation at this point in time.
I have heard that chorella
I have heard that chorella cannot be taken with a seafood/ iodine allergy? Is this the case with this formula? Would love to try it if I can!
Hi Courtney,
You are
Hi Courtney,
You are correct, the Baseline of Health Foundation is just a non-profit organization dedicated to offering free health information on natural remedies. The founder, Jon Barron, sells his formulas through Baseline Nutritionals: http://www.baselinenutritionals.com/shop.php?display=products
Hi there, just had blood test
Hi there, just had blood test results back with Vitamin D toxicity so I have too much calcium in my blood. The magnesium is low also. Therefore it could be related to that or I have been using a D3 supplement (not avidly or anything). My Vit D levels are at35nmol/L, the Dr hasnt phoned me with any worry so my assumption is it cant be that bad. I decied to increase Vit K by using miso soup and have felt much better the last few days. Lots of water to dilute the blood. My stomach pain have eased a lot. My question is…. Some studies are showing that the Vit K2 can also take the excess calcium outof solft tissue. So that sounds great but Miso also contains a good amount of calcium, so I didnt know if I may add to the problem. I was quite red in the face and this seems to have mainlu gone. However I am now wondering if this herbal chelation method would help also remove calcium that may have built up fromt he soft tissues. Its linked fromt he killer calcium page so I believe thats what yo are getting at, but its not mentioned in the above article. I have also bought some K2 in supplement form but am a bit worried again about taking supplements so thought th efood route was the best choice in the first instance?
Hi. I was wondering if your
Hi. I was wondering if your chelation system will help detox post mri. I’m very concerned about the contrast. Thank you sooo much for your response.
Metal Magic is made from just
Metal Magic is made from just cilantro and chlorella, so it appears those two ingredients are a problem for you.
Hi. I have manganese toxicity
Hi. I have manganese toxicity and am wondering if this formula would work on that. I’m also wondering if chlorella and cilantro can cross the blood-brain barrier. If so, is there any risk of moving lead or manganese from the soft tissue to the brain?
Thanks!
If you have a seafood allergy
If you have a seafood allergy, then it is likely you will have a problem with chlorella.
I’ve heard chelation helps
I’ve heard chelation helps Autistic children detox from heavy metals. Any restrictions or warnings or a way to do it safely?
I’m so glad Jon updated this
I’m so glad Jon updated this formula. I love his formulas, but I was one of the ones who experienced a problem with this one… (when I did his heavy metals cleanse, I could feel things “buzzing” around inside my head). Someone from Baseline Nutritionals told me it couldn’t be the formula because it only contained cilantro and chlorella and that I must have a problem with those two ingredients (which I’d never had–just with this particular formulation). Now I can give this formula another try!
The formula referred to in
The formula referred to in the article is for removing toxic heavy metals from the body. An MRI would have no impact on that.
Is there some way to test to
Is there some way to test to see if your body is needing to be cleansed of heavy metals and then to determine how long you should do it for?
This person was refering to
This person was refering to the Gadolinium that is in the contrast. Tests have shown that the Gadolinium deposits in all the organs and bone. There are millions of us with Gadolinium deposited in our bodies from MRI’s with contrast. Gadolinium is a Lanthanide rare earth metal.
Is there a detox formula for
Is there a detox formula for those of us allergic to cilantro and or chlorella? allergic to many foods including seeds, herbs, nuts, etc.
Manganese is a heavy metal.
Manganese is a heavy metal. It will be chelated along other heavy metals. As far as crossing the blood brain barrier, one of the key values of cilantro as a chelating agent is that it does cross the blood brain barrier and thus chelates heavy metals in the brain and then transports them out.
Hi Baseline 🙂
Hi Baseline 🙂
following the guidlines given in Jon’s book – ‘Lessons from…’; on page 106/7 he gives an outline of the cilantro pesto for heavy metal removal.
Have you any idea how long this (mixed with some chlorella) will keep in the fridge. Would i need to cut it into 3 seperate containers for example, freeze 2 of them, and thaw 1 of them at the end of each week?
Many thanks for your help 🙂
Will I be able to take my
Will I be able to take my medications during the 42 days of chelation?
You have me sold, but I can
You have me sold, but I can’t find the buy page.
I have read that Chlorella
I have read that Chlorella can increase sensitivity to the sun? For me this would be a turn off, is there any explanation for this?
I want to know more .what
I want to know more .what clean your blood
I had a similar question to
I had a similar question to Jess earlier. Does this work to remove calcium?
We don’t sell anything at the
We don’t sell anything at the Baseline of Health Foundation, we are a non-proft that gives free natural health information. See the link above called “Products.” All formulation recommendations and the companies that sell them are listed on that page.
For heavy metal detoxing, you
For heavy metal detoxing, you can try a formula based on EDTA. Also, don’t forget, the article above only talks about heavy metal detoxing. You want to consider a whole body detox. http://www.jonbarron.org/program/full-body-detox-program
I have successfully used many
I have successfully used many of the formulas developed and sold through Baseline Nutritionals, and greatly appreciate the expert and unparalleled advice that Jon Barron provides on this website.
I have a general question about the risk of all heavy metal detox formulas for people who have been diagnosed with leaky gut. At a recent seminar I attended for Hashimoto’s Thyroiditis, the presenter strongly discouraged the use of all such heavy metal detoxes because of the risk of re-absorption of the loosened metals back into the systems of people with leaky gut, and the resultant dangerous movement of those loosened metals into the brain, thus causing permanent damage. I am interested in hearing Jon’s take on this position. Thank-you.
There’s nothing special about
There’s nothing special about chlorella and sun sensitivity; it’s the chlorophyll in the chlorella. Chlorophyll heightens sun sensitivity in some people. If you have no problem eating high chlorophyll vegetables–such as spinach, kale, chard, collards, broccoli, grapes, celery–then it is unlikely chlorella will present a problem.
You should work with your
You should work with your doctor if on any medication and considering detoxing, but for most medications, it should be fine.
Check with an alternative
Check with an alternative health practitioner. The standard methods for determining heavy metal toxicity are blood testing, urine provocation testing, and hair analysis—and usually a combination of all three.
I am trying to detox from an
I am trying to detox from an Injection of botulinium toxin.
Will choorella and cilantro work for me..
Thank you
When formulating Metal Magic
When formulating Metal Magic were clinical studies done on the following heavy metals: Cesium, Palladium and Platinum? I am elevated in each of the metals in addition to Lead and Mercury. Do heavy metals work synergistically in the body?
Thanks.
If a person is now taking
If a person is now taking meds for hypertension .. and he decides to use protolytic enzymes, would its use remove some of the effects of the medication within the arterial wallls ?
If so, how would you minimise any adverse results ?
Thank you for any information
Ed R
If the kidneys and liver are
If the kidneys and liver are somewhat compromised, can using your detox formula actually cause more damage due to the organs being unable to handle the newly-active toxins in the bloodstream? Should these organs be healed as much as possible first, before the detox? Thanks, TD
Unlikely. They are heavy
Unlikely. They are heavy metal chelators.
Calcium is technically a
Calcium is technically a heavy metal. “Heavy metals” is an inexact term used to describe more than a dozen elements that are metals or metalloids (elements that have both metal and nonmetal characteristics) This includes Calcium, Copper, Lead, Arsenic, Cadmium, Chromium, Mercury, and Manganese. Because they cannot be degraded or destroyed, heavy metals are persistent in all parts of the environment.
The cutoff point to be considered a heavy metal is a specific gravity of 5 gm per cubic centimeter. With that in mind, the specific gravity of both calcium is above 8 — thus it is a heavy metal. Note: with a specific gravity of 2.55-2.80, aluminum is not a heavy metal — but is treated as such in terms of toxicity and will also be pulled from the body by a good chelating agent.
That said, an oral chelating formula can be highly effective at removing small amounts of heavy metals which have great toxicity at very low levels – mercury, arsenic, nickel, cadmium, etc. It will not remove a significant enough amount of metals that have a substantial presence in the body, such as calcium or iron, to be even the slightest bit noticeable. Also, the important thing with calcium is not so much the amount you consume or chelate, but how your body regulates where it redistributes the vast amount that is already present in your calcium bank—your bones—and for that, body pH is a primary consideration.
Hi, has Cilantro essential
Hi, has Cilantro essential oil the same effect? It is stronger or it has lost its detox properties. In others sites, (sellers mostly) say it does but there is not any scientific bibliography. Could you tell me something about? I have Cilantro essential Oil but I have never use it to ingest or to massage. Thank you.
I mistakenly took 5000 Vit B
I mistakenly took 5000 Vit B 12, as part of improving over all health after Chemo and Radiation.
Unbeknown to me there was high levels of Nickel in this product, and am so allergic to Nickel, if something just sits on my skin for five minutes with Nickel I will have an immediate reaction.
For the past three weeks I am crawling out of my skin with itch, and breaking out where ever there is a hot spot….ie bra, panty creases in my legs and under arms…
Do you recommend your product for detoxing….
Hi, I need to know if this
Hi, I need to know if this would work on children ages 6 to 13 years? Since I’ve known they absorb heavy metals in a greater way that us adults.
We would question whether a
We would question whether a cilantro essential oil is concentrated enough in the bioactives that actually break the bonds heavy metals have with human tissue to be effective as a detox agent. A quality tincture, on the other hand, contains all bioactives in a concentrated form and has proven to be effective.
Proteolytic enzymes are
Proteolytic enzymes are unlikely to change the efficacy of any hypertension medications, be they diuretics, calcium channel blockers, or beta blockers. However, using proteolytic enzymes might require a change in the amount of medication you need. In any case, you will want to consult with your physician before using any supplements.
Will this help remove all
Will this help remove all Platinum group metals from the body?
I wonder if you took
I wonder if you took cyanocobalamin or methylcobalamin.
Avoic the cyano form. Cyano is cyanide-based.
A different brand might not have the Nickel in it.
Yes, it would probably work
Yes, it would probably work with children the same as with adults as the mechanisms of action would remain unchanged. That said, we recommend that you consult with your pediatrician before using any herbal formulas with children. This advice would hold true for any natural formula that actually works VS a placebo.
Should Metal Magic be taken
Should Metal Magic be taken away from mealtimes? I’ve been using chlorella and modified citrus pectin for lead chelation until now, and that’s taken half an hour before a meal. That’s so the pectin, and maybe the chlorella, can act as a bile-toxin binder. Does Metal Magic have a similar relationship with the release of bile? Thanks for answering questions!
I have ALS . I was tested for
I have ALS . I was tested for metals after a chelating process of EDTA 1500 mg thru IV over a 3 hour period. then they tested my urine specimens over the next 6 hours. The test came back and my cadmium was 5.8 and lead was 17. They were in danger zone. Also had nickel thorium and tungsten, but these were high but not in danger zone. My question is how much of s your product would I need to be equivalent to 1500 mg of EDTA. The IV is expensive and I’m trying to find another option to remove toxins in my body.
The formula has never been
The formula has never been specifically tested for platinum, but the combination of cilantro and chlorella should work for all heavy metals.
Hi, can you please tell me if
Hi, can you please tell me if the ‘Metal Magic’ would help a child of 7 with Autism, or what would you recommend should be undertaken. Currently a MMS formula by Jim Humble is being administered with positive but slow results, but tastes so vile for the child. I’m hoping to find something else more palatable and possibly even better/quicker acting. Thank you.
Hi, is this suitable for
Hi, is this suitable for children? and what will be the dosage?
Instead of doing the initial
Instead of doing the initial 6 bottle cleanse back to back, could I spread it out over 6 months by taking one bottle every month? Would it still have the same effect?
I haven’t read all the
I haven’t read all the questions about this article, so this may have been answered.
I’m wondering if there are any physical signs of detoxing using Metal Magic. Perhaps the urine changes odor, or some skin change.
Thanks
Good evening,
Good evening,
Do you think this could help for nickel allergy?
Regards
My son is 7yrs old and is on
My son is 7yrs old and is on the autism spectrum with a lot of behaviour issues and is quite hyper active.he has been tested and discovered heavy metals, lead, mercury, aluminium, arsenic. please can i do the cilantro and chorella detox for him, and how do i do it, should it be in the form of a smoothie glen with some fruits to sweeten it. kindly advise
Jon, I have been using many
Jon, I have been using many of your products for years. Colon cleanse and colon corrective may have save my life and your women’s formula helped me tremendously, particularly through menopause. Starting on the Phi-zymes, loads of research great research on this. I very much appreciate your sharing of knowledge as well as high quality products. Twelve years with you and still here with good quality of life Jon. Thanks for the help!
This product sounds amazing.
This product sounds amazing. Can you tell me where the Chlorella is sourced and is it tested for heavy metals? I am very cautious of Chlorella after the amount of radiation that was and is still being emitted from Fukushima into the ocean and atmosphere.
I have tested high for nickel
I have tested high for nickel and moderate for lead. Will this formula work to eliminate these metals? Thank you!
HI
HI
Where do I order the heavy metal detox formula. I want to order it NOW. Also can my cats take it? They also were exposed to chemicals in the environment and pesticides.
I’d like to talk to you about these things as I have now brain toxicity and more issues. Now I can’t be near any kind of paint or cleaners if they aren’t 100 percent non toxic non chemical. I get reactions right away making it hard for me to live anywhere now.
Does this work getting rid of
This works getting rid of toxins from vaccinés
Where do I buy this product to get rid of toxins